Cart is empty
CosmeticTattoo.org has Retired
28/02/2023
by Andrea Darby - Master Medical Tattooist & Industry Educator
After more than 10 years of providing free education and marketing to the global Cosmetic & Medical Tattoo industry CosmeticTattoo.org has retired today, read on to find out more.
▼ Continue Reading ▼
|
In 2012 I provided a lecture and demonstration to over 500 industry members and
medical practitioners on scalp micropigmentation at the Permanent International
Congress in Berlin Germany, to say that there was strong interest would be an
understatement.
After the presentation I was inundated with questions and requests from
attendees and other presenters many of whom described their frustrations in
gaining access to reliable information and their concerns that there was always
a high price tag attached to any promotional avenue that was available to them
at that time. In the weeks after the congress I continued to receive hundreds of
emails from cosmetic tattoo technicians from around the globe as the word spread
about my presentation from the attendees to others in their home countries. My partner Derek & I discussed the challenges that had been raised by
industry members and after a lot of brainstorming with our advisory network we
decided to launch CosmeticTattoo.org a place where cosmetic and medical
tattooists could promote their brand and gain access to high quality educational
information free of charge on a platform that we created and the first of its
kind for our industry. I am proud to say that in a little over 10 years
CosmeticTattoo.org has provided over 32,000 client referrals to our
international members and has answered nearly 85,000 enquires by clients and
cosmetic tattoo technicians. Our
Educational Articles became so popular that we also had to contend with
preventing plagiarism and copyright infringement by
other cosmetic tattoo industry members, magazine publishers, associations,
manufacturers and
even a few health professionals. As the saying goes "imitation is the
highest form of compliment". Fast forward beyond the pandemic to 2023 and like many people we have been
re-evaluating and prioritizing, CosmeticTattoo.org has been a fantastic project
and we have met, corresponded with, and helped thousands of people. After 10
years of providing a free platform for the global industry we think that we have
contributed more than our fair share and felt it was time to retire the Org and focus our
time on other projects. When I told some of our members that CosmeticTattoo.org
was going to retire the first thing all of them said was what about the
articles? Don't worry we have preserved our
Educational Articles and our
Science
Library right here at CTshop.com.au under
Our Publications within
our
new e-commerce store. To all of our past members thanks for being part of CosmeticTattoo.org. Andrea Darby (the first Global Membership platform for Cosmetic & Medical Tattooists and Micropigmentation Specialists)
Date of most recent revision:
28/02/2023 (mutatis mutandis) Copyright © 2023 CTshop.com.au & the article author All Rights Reserved. No copying, transmission or reproduction of site content is permitted without our prior written consent.
Printing Restriction: This article is print disabled, please read our Intellectual Property & Copyright Policies if you would like to request a copy or permission to use the article content for any purpose. |
Site News Selection
Educational Article Selection
Regulatory Article Selection
Client Case Studies Selection
Science Library Selection
Complete regrowth of hair following scalp tattooing in a patient with alopecia universalis
31/01/2023
Atypical Intraepidermal Melanocytic Proliferation Masked by a Tattoo: Implications for Tattoo Artist
20/09/2018
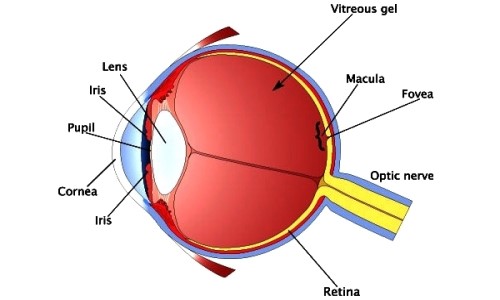
Chemical conjunctivitis and diffuse lamellar keratitis after removal of eyelash extensions
26/08/2018
Scarless Breast Reconstruction: Indications and Techniques for Optimizing Aesthetic Outcomes
07/04/2018
High speed ink aggregates are ejected from tattoos during Q‐switched Nd:YAG laser treatments
28/03/2018
Unveiling skin macrophage dynamics explains both tattoo persistence and strenuous removal
08/03/2018
Granulomatous Tattoo reaction with Associated Uveitis successfully treated with methotrexate
08/02/2018
Identification of organic pigments in tattoo inks & permanent make-up using laser mass spectrometry
07/02/2018
Microbiological survey of commercial tattoo and permanent makeup inks available in the United States
03/02/2018